
Please come hear my presentation about BRG Therapeutics at the Live Pitch Event hosted by Devin Thorpe of SuperPowers 4 Good. See News & Events for details.

The Unmet Need:
Each year in the U.S., over 600,000 people die due to cancer. The majority of these deaths are due to the unresolved problem of drug resistance. Most fatal cancers are drug resistant.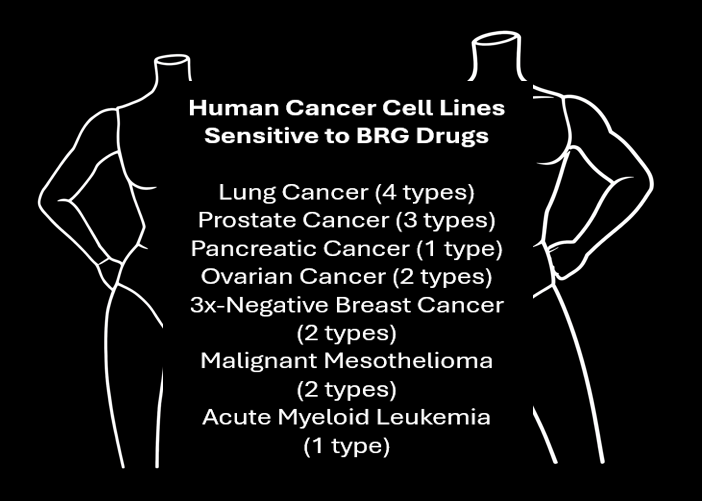
Our Solution
Based on our laboratory data, BRG drugs work in human cancer types that have few to no effective treatments, and they alter tumor cell resistance to selected anticancer agents so that all to nearly all tumor cells are killed after just one exposure when a BRG drug is used in combination with a second anticancer agent.